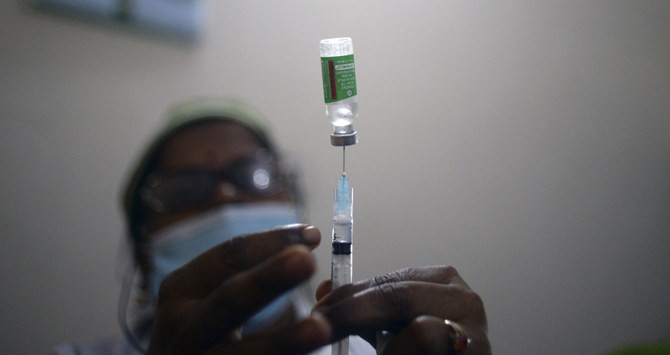
DHAKA: Bangladeshi authorities have conditionally cleared the country’s first coronavirus vaccine for clinical trials, which the producer expects to complete in the next few months.
The vaccine, Bangavax, is a new generation mRNA vaccine that, like the Pfizer and Moderna ones, teaches our cells how to make a protein that triggers an immune response inside our bodies. Developed by Dhaka-based Globe Biotech Ltd. (GBL), the vaccine was approved for production by the country’s drug regulator in late December.
On Wednesday, the Bangladesh Medical Research Council (BMRC) approved clinical trials of Bangavax under the condition that “before starting any human trial, the vaccine producing company needs to conduct an animal trial on monkeys or chimpanzees,” BMRC director Prof. Dr Ruhul Amin said.
GBL has been waiting for the trial approval since January.
“It’s a lengthy process,” Amin said. “However, we are doing our best to facilitate the trials of Bangavax.”
Dr. Mohammed Mohiuddin, head of quality at GBL, said that while the company is now waiting for the BMRC’s written recommendations, it is preparing to start the trials.
“It will take us eight to nine months to complete the whole process,” he said. “Since we are using pure mRNA technology in Bangavax and no virus is used in this process, we are supposedly not required to make an animal trial.” He said that GBL was in touch with organizations abroad as there is no institution conducting animal trials in Bangladesh.
“To run an animal trial, some foreign companies are asking for a G2G — government to government contract. We hope the government should extend help to us in this case,” Dr. Mohiuddin said.
As Bangavax is estimated to cost $10-$15, several dollars cheaper than the Pfizer and Moderna vaccines, it may help Bangladesh with its immunization drive, in which only 2.6 percent of the country’s 166 million people has been vaccinated so far, mainly due to a shortage of COVID-19 jabs.
FASTFACT
Dr. Mohammed Mushtuq Husain, an adviser at the state-run Institute of Epidemiology Disease Control and Research (IEDCR), said if Bangavax trials prove successful they would position Bangladesh ‘ahead in the vaccine race amid this global crisis period.’
GBL says it has the capacity to produce 10 million doses a month, and its lab tests on mice suggest that one dose would suffice.
“We are expecting that it will be a single dose vaccine as we found about 100 percent efficacy rate during lab trial on mice,” Dr. Mohiuddin said.
Dr. Mohammad Mushtuq Husain, an adviser at the state-run Institute of Epidemiology Disease Control and Research (IEDCR), said if Bangavax trials prove successful they would position Bangladesh “ahead in the vaccine race amid this global crisis period.”
“They (GBL) should be provided with necessary administrative and financial support as and when required. But the highest level of precaution is a must at every stage of the trials,” he said.
“If we become successful in this endeavor, Bangladesh may consider exporting vaccine to other developing countries after meeting local demand.”